the little things that run the Earth
by Lauren J. Bryant
From clams and climate to human health, microbes rule.
The esteemed biologist E. O. Wilson once said, when describing the world’s biological diversity, “It’s the little things that run the Earth.” Microbiologist Barbara Campbell certainly agrees, but she leans toward the description from Dr. Seuss:
From near to far,
from here to there,
funny things
are everywhere.
Campbell, an assistant professor in the Department of Biological Sciences, is a microbial ecologist—the funny things that occupy her scientific mind are microbes, the world’s abundant microscopic organisms that include algae, bacteria, and viruses. Campbell focuses mainly on bacteria found in marine environments, especially in estuaries—where river and ocean meet—around the Delaware and Chesapeake bays.
Why this fixation on little, funny things?
“Microbes rule!” Campbell exclaims. “They’re everywhere, and we can’t live without them.”
We can’t live without them because microbes play essential roles in everything from regulating human health (think of the gut microbiome) to sustaining the natural cycles on which the life of plants, animals, and the earth’s entire biosphere depend. In short: Little things add up, big time.
“I tell my students, ‘What’s great about microbes is they are so diverse and can do so many different things,’” Campbell continues. “‘And what’s bad about microbes is they are so diverse and can do so many different things.’ Microbes are hard to study because they are so small and so diverse, but that diversity also makes them powerful.”
Campbell’s own description of how she accomplishes the difficult study of microbes has a Seuss-like simplicity. “I look at who they are and what they’re doing,” she says. The reality is a bit more adventurous and high tech.
On deck
Campbell (along with her frequent collaborator, David Kirchman, of the University of Delaware) takes two main approaches to investigating the behaviors of bacteria in coastal estuaries. First, she spends a fair amount of time on deck—on ship decks, that is, such as that of the R/V Hugh R. Sharp, a coastal research vessel in the University-National Oceanographic Laboratory System fleet whose home port is at the University of Delaware.
During research cruises, the scientists and students aboard collect samples twenty-four hours a day, typically every three to four hours, with the goal of learning how bacterial activities change over the course of a day and at different water salinities. They conduct experiments and analyses using the ship’s laboratories. In between the sampling and analyzing, Campbell says, the researchers “read, talk science, or head up to the bridge to get a great view of the water.”
Campbell has been out to sea five or six times since 2011 to conduct microbial research in estuaries. Her most recent trip occurred in late October 2014. “That one was a little rough,” she says. “A nor’easter was coming up along the coast, so we had to not do some things we’d planned on.”
These sea voyages, rocky or no, are necessary, Campbell explains, because “about ninety-nine percent” of microbes cannot be isolated and grown in a typical laboratory setting. It’s just not possible to replicate the exact requirements for the microbes’ growth—their necessary nutrient sources, for example, or the optimal temperature. And even if the right conditions could be created, artificially grown microbes would still not be preferred because they would be outside of their natural environment. That natural environment is important, Campbell says, because microbes are “a lot like us.”
“Microbes can’t live alone, typically,” she says. “They’re like us; we’re always interacting with other people. Microbes interact with other microbes, for food sources and such. But there’s a lot of interaction between microbes that we just don’t recognize yet.”
A universe in a drop of water
On dry land, Campbell and her students study the water samples collected at sea using the methods of metagenomics and metatranscriptomics. Essentially, metagenomics is the study of the genetic material from a community of uncultured microbes (microbes taken directly from their environment). Metatranscriptomics refers to the study of RNA molecules in groups of interacting organisms.
“We sequence all of the genes in all of the microbes in our sample,” she says, “and then we go back and figure out what those genes are. We can determine the types of bacteria that way. We determine who’s there by looking at what genes they have.”
Next, Campbell looks at what the bacteria are doing. Currently, working with a grant from the National Science Foundation, Campbell and her colleagues are looking at bacterial growth rates. To date, Campbell has found that the different microbes vary their activity and growth. Contrary to what scientists predicted, some abundant bacteria in estuary environments are not very active, while other, rarer bacteria are growing, and faster than expected.
What do these faster growth rates mean? Campbell has lots more data to analyze before she can answer that question with specificity, but there’s no question that this kind of investigation into microbial activity is significant.
That’s because, as Campbell says, microbes rule. Quite literally. Microbes dominate, inside our bodies and out. Scientists use various calculations and comparisons to convey the vast number of bacteria that exist—more than 1030, some say (inside the human body alone, there are 10 times more bacterial cells than human cells), or more than all the stars in the Milky Way. Campbell uses the familiar context of the ocean. Every milliliter of seawater—about 1/5 of a teaspoon—contains somewhere around 1 million microbes, she says, and “oceans average about four kilometers [a little over two miles] in depth and cover seventy percent of the Earth’s surface. So, if there are a million microbes per milliliter. . . it’s very hard to comprehend.”
 An array of sampling equipment called a rosette is lowered into the Delaware Bay. Image courtesy of Mason Jones.
An array of sampling equipment called a rosette is lowered into the Delaware Bay. Image courtesy of Mason Jones.Cycles of life
Beyond their sheer numbers, these countless microbes also play essential roles in keeping our world going. Microbes are the linchpins of Earth’s biogeochemical cycles, a term that generally refers to the circulation of elements from Earth to atmosphere and atmosphere to Earth. Think of the carbon cycle, for example: Plants absorb carbon dioxide from the atmosphere and release oxygen through photosynthesis; organisms, including humans and bacteria, consume carbon for energy, then return the carbon to the atmosphere when they breathe and to the ocean and earth when they die and decompose.
As the National Research Council puts it in its publication The New Science of Metagenomics: Revealing the Secrets of Our Microbial Planet, “microbes are essential for every part of human life—indeed all life on Earth . . . The chemical cycles that convert the key elements of life—carbon, nitrogen, oxygen, and sulfur—into biologically accessible forms are largely directed by and dependent on microbes.”
So what happens when something disrupts these trillions of bacteria carrying out essential processes on which all life depends? That’s what Campbell wants to find out.
The bacteria that Campbell studies consume organic matter, just like humans do, and respire, or release, carbon dioxide. Understanding their varying growth rates is important, she says, because the faster microbes grow, the more they contribute to biogeochemical processes.
Campbell hypothesizes that rates of bacterial growth are sensitive to different environmental conditions, such as changes in the amount of light, temperature, or nutrients in the water. When there are more nutrients in the water from land runoff and when the water is warmer, she explains, the bacteria are more active and consume more carbon before the carbon can sink to the bottom of the ocean. And the more carbon the bacteria consume, the more CO2 they release, she says, “which is going back up into the atmosphere.”
If the impact of an invisible microbe releasing a bit more CO2 seems trifling, just recall the numbers: 5 million microbes in a single teaspoon of seawater. With Campbell’s more accurate estimates of “who they are and what they’re doing,” especially how environmental changes affect bacterial activity, her basic-science research will enable others, she says, “to better estimate outcomes under different environmental conditions including future climate change that may involve dramatic swings in water availability through droughts and floods, sea level rise, and increasing temperatures.”
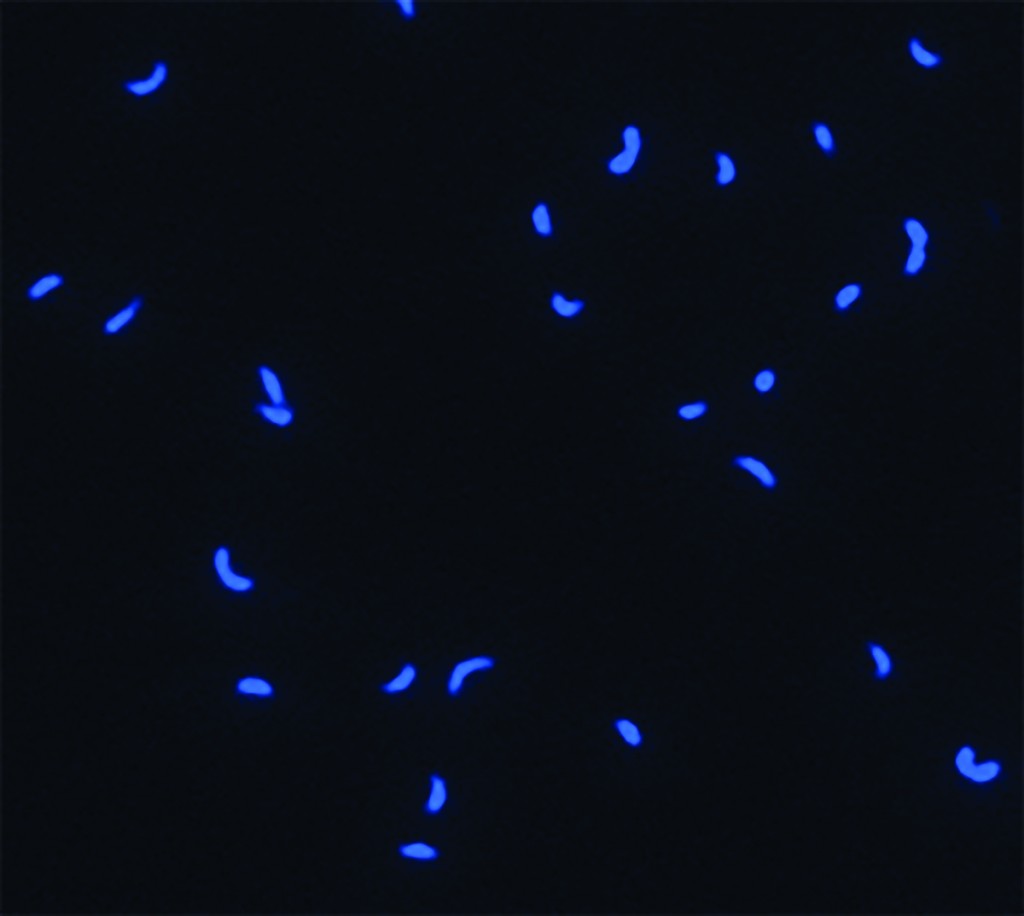
Pelagibacter ubique, one of the most abundant microbes in the ocean, magnified about one thousand times and stained blue. Image courtesy of Thomas Lankiewicz and Matthew Cottrell, University of Delaware.
Cows and clams
Campbell’s study of bacterial growth rates is just one of the ways she and her lab partners explore how tiny bacteria have huge effects on human health. In a related study that also uses samples collected from coastal and bay waters, Campbell is evaluating water quality, particularly levels of fecal contamination. The FDA uses levels of the bacteria Enterococcus as an indicator of water quality, Campbell explains. Enterococcus is typically found in human intestines and feces; the problem is, she says, it’s also found in a wide range of animals, not just in humans.
“The bacteria can come from cows, horses, pigs, wild animals, birds, as well as humans,” she says.
That makes it difficult for state and federal agencies to pinpoint where contamination may be coming from, so Campbell is applying gene-sequencing technologies and statistical analyses to water samples to help answer that question.
“We can make inferences from the genetic data about which bacteria come from humans, cows, etcetera,” she says. “It’s important to know where the contamination is coming from because then we can say, this is normal contamination, or it’s from human sewage. That would allow the state to figure out how to best address a problem.”
Likewise, Campbell is exploring how bacteria hanging out in the gills of clams influence the condition of vital coastal ecosystems. In a study funded by the NSF’s Dimensions of Biodiversity program, Campbell collaborates with Annette Engel, of the University of Tennessee, and Laurie Anderson, of the South Dakota School of Mines and Technology, to consider the impact of environmental changes on lucinids, a common species of clam found in Southern coastal marine environments.
The clam’s gills contain symbionts, bacteria that live in mutually beneficial relationships with the clams and the seagrass beds that the clams inhabit. As part of its natural growth process, Campbell explains, seagrass produces sulfide (a potentially toxic chemical compound containing sulfur). The symbiotic bacteria use the sulfide to turn carbon dioxide into sugars, and the sugars are used by the clams for growth. This three-way process is integral to the health and well-being of the seagrass beds. If sulfide levels get too high, the seagrass stops flourishing.
Working with the symbiotic bacteria, Campbell is again reading genetic material in the samples to see who’s there.
“We’re looking at the variation in types of microbes present in seagrass beds versus non-seagrass bed environments as well as what the symbionts are actually doing and how they are contributing to what types of processes,” she says.
Like her research on bacterial growth rates in estuaries, understanding the activities of an invisible organism in maintaining the seagrass environment can have huge benefits. Seagrass beds act as marine “nurseries,” Campbell notes, harboring all manner of small fish and other sea creatures. Maintaining the health of seagrass beds is critically important to the entire ecology of coastal systems.
 Members of the scientific crew aboard the R/V Hugh R. Sharp. Image courtesy of Michael Gonsior, University of Maryland.
Members of the scientific crew aboard the R/V Hugh R. Sharp. Image courtesy of Michael Gonsior, University of Maryland.About the unknown
Despite the power and plentitude of microbes, scientists still know very little about them, although that’s changing fast. Until about ten years ago, the scientific technologies needed to support the kind of research Campbell is carrying out didn’t exist. Since then, she explains, next-generation gene-sequencing technologies have emerged, which allow genetic information to be determined in much greater detail at far faster speeds and much less cost. In addition, new supercomputers, such as the high-performance Palmetto Cluster at Clemson, and new informatics technologies now enable researchers to make sense of the massive amounts of data being generated.
For many years, Campbell says, because of past limitations on culturing and sequencing, scientists thought a milliliter of water contained only about a thousand species of bacteria, because that’s all that could be cultured and identified. Now, says Campbell, “we know there are a million bacteria in that ml of water, and they are not all of the same type. And the next ml might have some of the same bacteria, but it always has new ones too. It’s a very diverse environment, so we have to sequence a lot to figure out who’s there. And as the technology gets more precise, we’ll find more and more.”
You might say all those millions of microbes give Campbell job security. There are many unanswered questions about how microbes work, which is fine with Campbell. She’s an active collaborator; she knows how to get her research funded (her formula: “Learn how to structure your science, then sell the importance of your work in answering things that other people need to know, then follow up and do the work.”). Most of all, though, she really, really likes asking questions.
“Just ask my mom,” Campbell says with a rueful laugh. “As a kid, I was always asking questions.
“I’m just interested in the unknown,” she continues. “I like asking questions about a lot of different things, and I can ask those questions in a lot of different ways.”
As Dr. Seuss put it: “Think and wonder, wonder and think.”
 Sunset from aboard the R/V Hugh R. Sharp as the team takes samples in the Delaware Bay near Delaware City in October, 2014. Image courtesy of Mason Jones.
Sunset from aboard the R/V Hugh R. Sharp as the team takes samples in the Delaware Bay near Delaware City in October, 2014. Image courtesy of Mason Jones.Barbara Campbell is an assistant professor of biological sciences, College of Agriculture, Forestry, and Life Sciences. Lauren Bryant is a writer living in Bloomington, Indiana.